A monotectic reaction consists of change from a liquid and to a combination of a solid and a second liquid, where the two liquids display a miscibility gap. A peritectoid reaction is a peritectoid rection, except involving only solid phases.  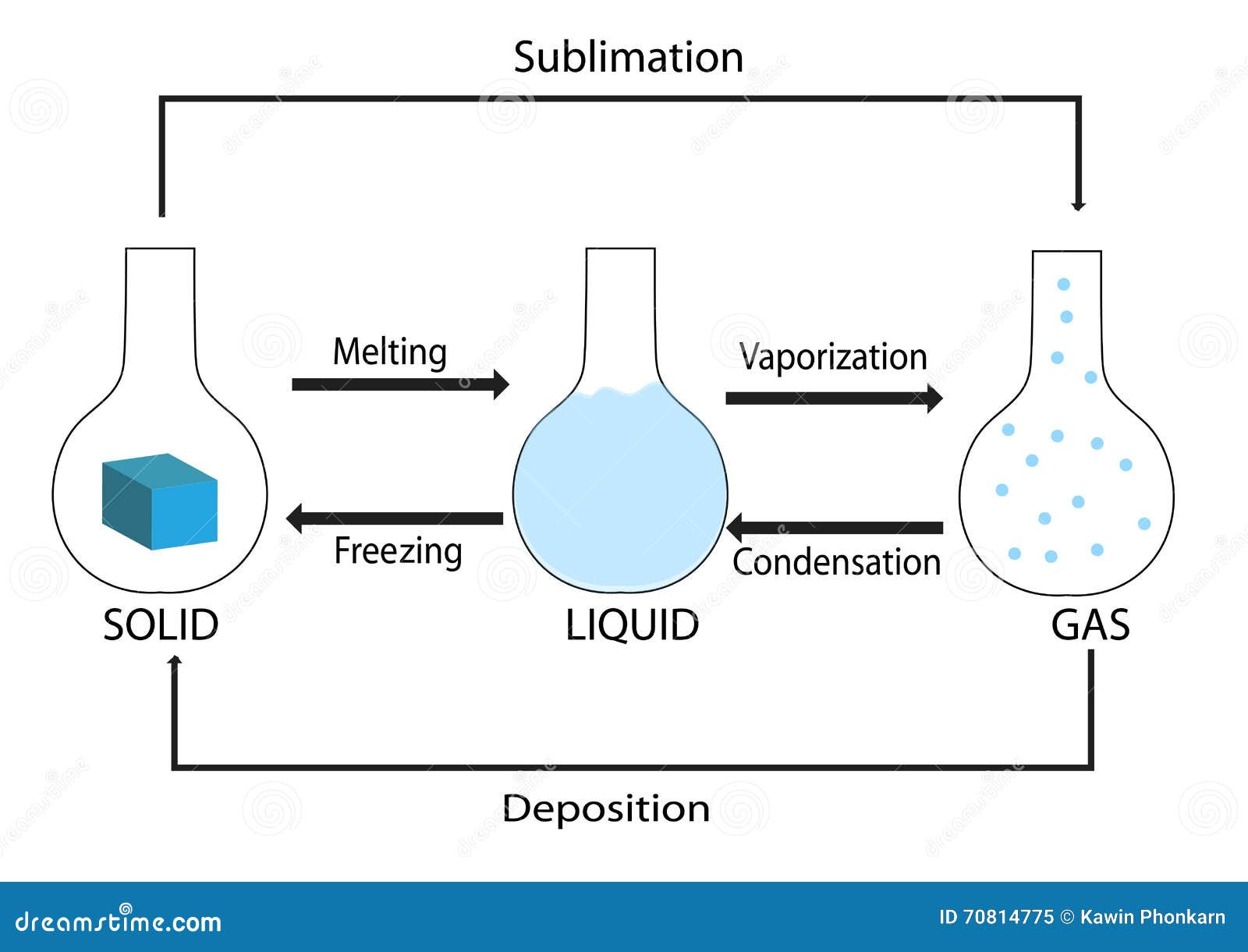
A peritectic transformation, in which a two-component single-phase solid is heated and transforms into a solid phase and a liquid phase. 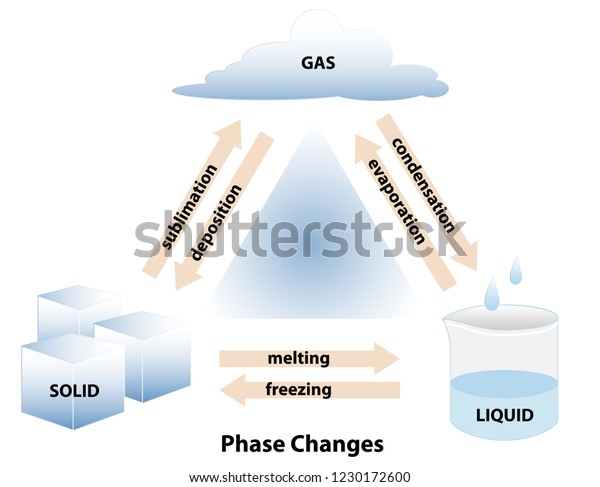
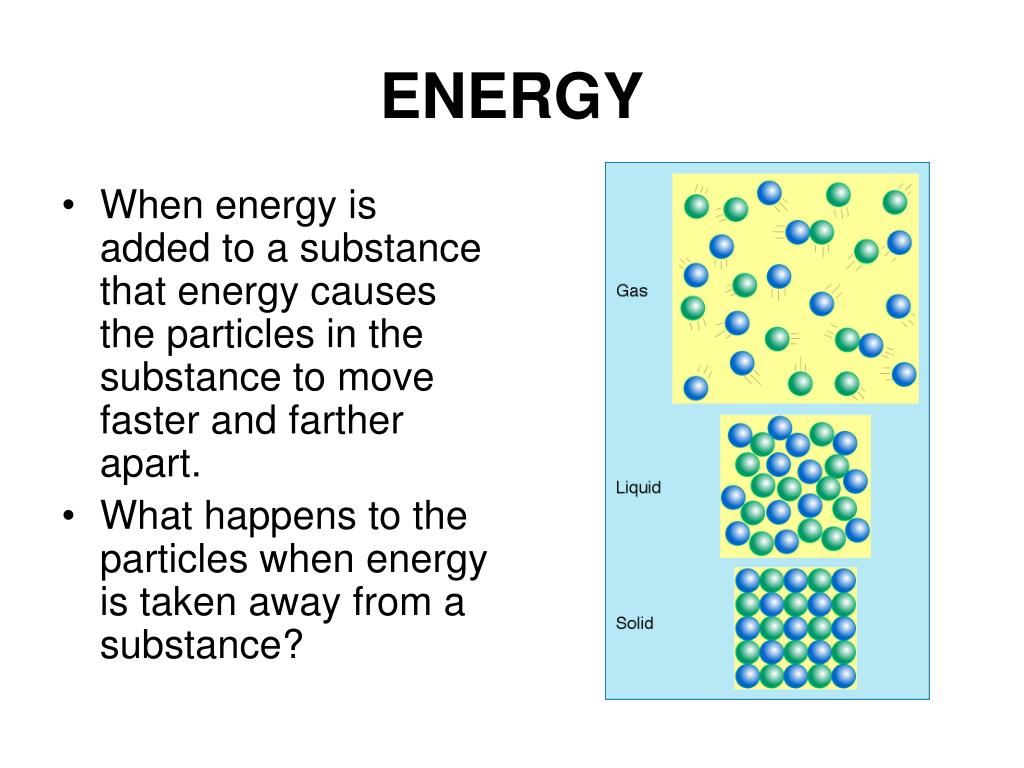
The same process, but beginning with a solid instead of a liquid is called a eutectoid transformation. There are also a number of phase transitions involving three phases: a eutectic transformation, in which a two-component single-phase liquid is cooled and transforms into two solid phases. This is often the case in solid solutions, where the two components are isostructural. While chemically pure compounds exhibit a single temperature melting point between solid and liquid phases, mixtures can either have a single melting point, known as congruent melting, or they have different solidus and liquidus temperatures resulting in a temperature span where solid and liquid coexist in equilibrium. Phase transitions involving solutions and mixtures are more complicated than transitions involving a single compound. The transition between differently ordered, commensurate or incommensurate, magnetic structures, such as in cerium antimonide. The transition between the ferromagnetic and paramagnetic phases of magnetic materials at the Curie point. A metastable polymorph which forms rapidly due to lower surface energy will transform to an equilibrium phase given sufficient thermal input to overcome an energetic barrier. As with states of matter, there are also a metastable to equilibrium phase transformation for structural phase transitions. Order-disorder transitions such as in alpha- titanium aluminides. The martensitic transformation which occurs as one of the many phase transformations in carbon steel and stands as a model for displacive phase transformations. The change from one crystal structure to another, from a crystalline solid to an amorphous solid, or from one amorphous structure to another ( polyamorphs) are all examples of solid to solid phase transitions. In elements, this is known as allotropy, whereas in compounds it is known as polymorphism. Phase transitions can also occur when a solid changes to a different structure without changing its chemical makeup. Metastable states do not appear on usual phase diagrams. This occurs in superheating, supercooling, and supersaturation, for example. The resulting state is metastable, i.e., less stable than the phase to which the transition would have occurred, but not unstable either. In exception to the usual case, it is sometimes possible to change the state of a system diabatically (as opposed to adiabatically) in such a way that it can be brought past a phase transition point without undergoing a phase transition. A phase transition usually occurs when the pressure or temperature changes and the system crosses from one region to another, like water turning from liquid to solid as soon as the temperature drops below the freezing point. Such a diagram usually depicts states in equilibrium. For a single component, the most stable phase at different temperatures and pressures can be shown on a phase diagram. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
